14 Aug 2020
The 6 Key Aspects of Analytical Method Validation
Analytical method validation is critical if our measured values are to have any worth. It can be a daunting task for the inexperienced and can add significant time and cost to analysis but if we look bigger, the time and cost of re-running or redesigning a method would pale in comparison (not to mention the cost of any damage caused by incorrect results).
Validation comes in many guises and what rules and requirements are followed will be dependant upon the constraints of the particular analytical problem the method is trying to solve. The aim is to try and get as much information from the lowest cost of time or investment whilst following a specific set of guidelines from a governing body such as ICH, MHRA or UKAS.
Although specific requirements may be different, the guiding principles are the same in that the method must meet six criteria for it to be considered “fit-for-purpose” and a handy mnemonic to remember they is:
Silly - Analysts - Produce - Simply - Lame - Results
Specificity - Accuracy - Precision - Sensitivity - Linearity - Robustness
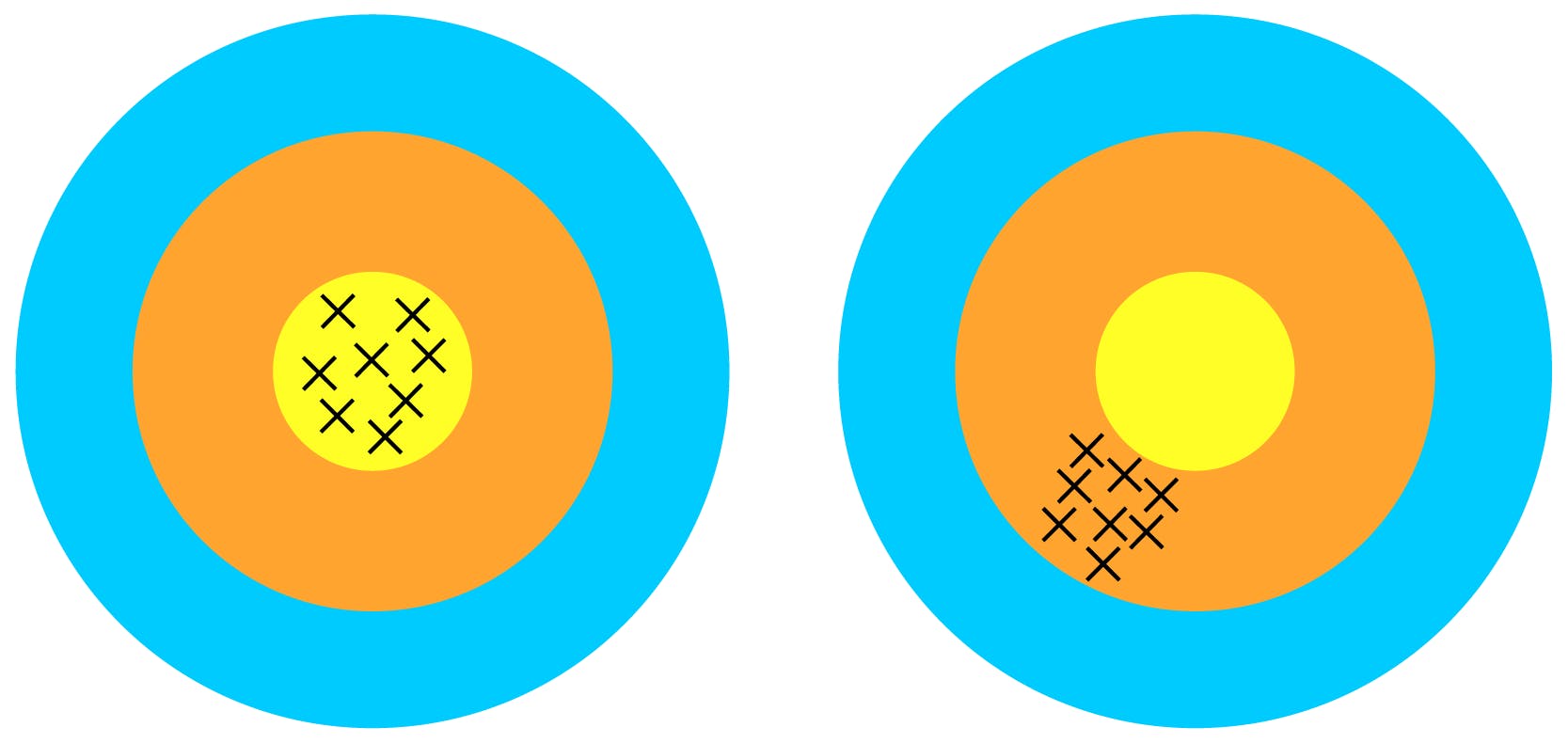
Specificity
Specificity is the ability to assess unequivocally the analyte in the presence of components which may be expected to be present. Typically these might include impurities, degradants, matrix, etc.’
The method needs to avoid as many false positives as possible as the target may be one of many components in a sample and specific method should only yield results to the target and the target only. A specific result is one that is free from interference.
Typically this is tested first because we need to make sure we are testing the right thing!

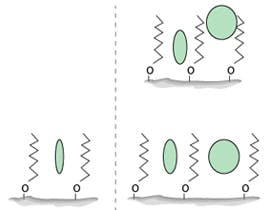
Purple = Positive result, yellow = Negative result. Non-specific method: rectangle included, specific method= only squares included.
Accuracy
‘The accuracy of an analytical procedure expresses the closeness of agreement between the value which is accepted either as a conventional true value or an accepted reference value and the value found. This is sometimes termed trueness.’
An inaccurate method delivers results which are not close to the true result. Accuracy is tested by preparing samples of known concentration, testing them and seeing how close the method result is to the known true value.
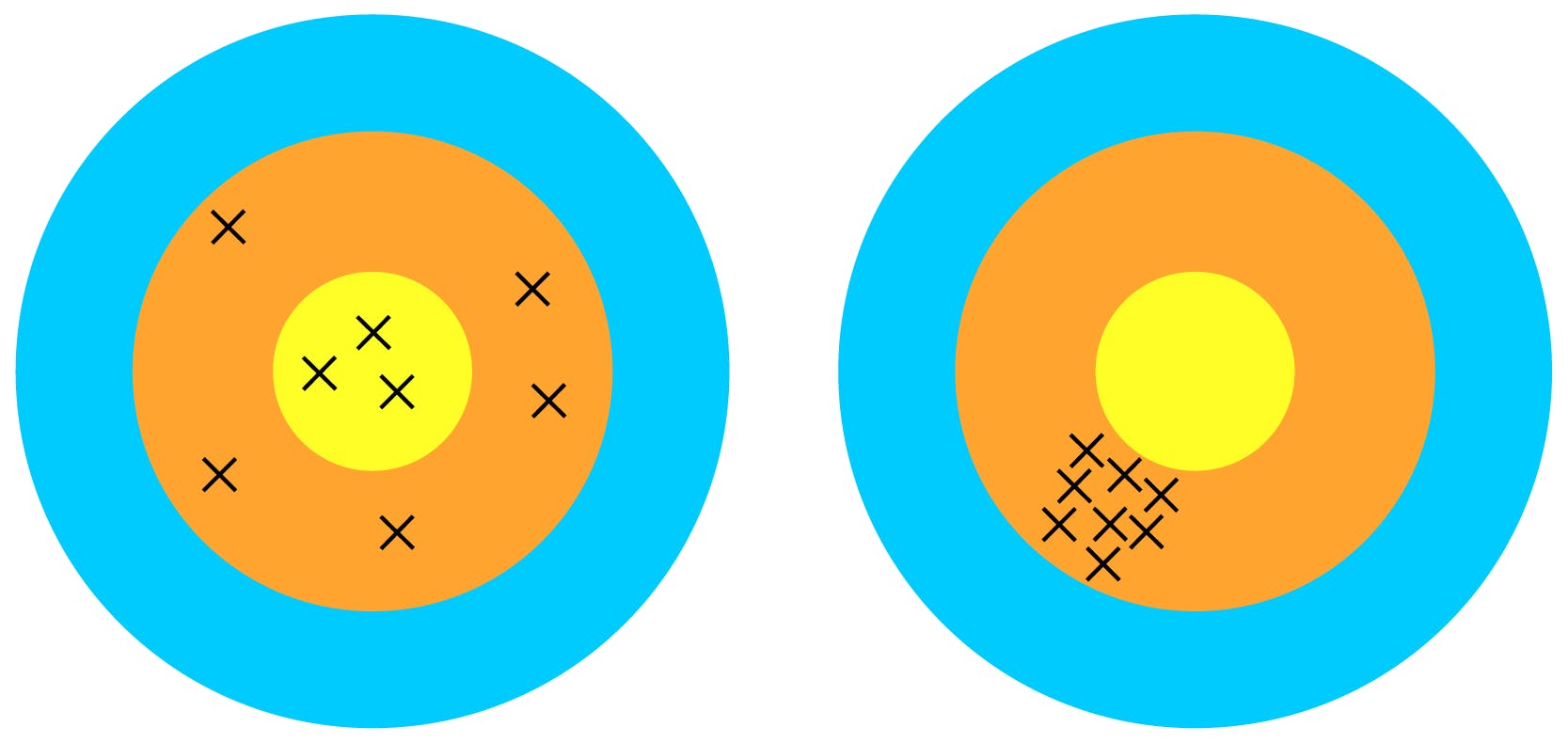
Precision
‘The precision of an analytical procedure expresses the closeness of agreement (degree of scatter) between a series of measurements obtained from multiple sampling of the same homogeneous sample under the prescribed conditions.’
If you repeat a measurement on the same sample, you should obtain the same result. The closeness in agreement of multiple measurements is considered precision. The method should be controlled in such a way that random factors are minimised and the response is due to the presence of the analyte only so it varies as minimally as possible.
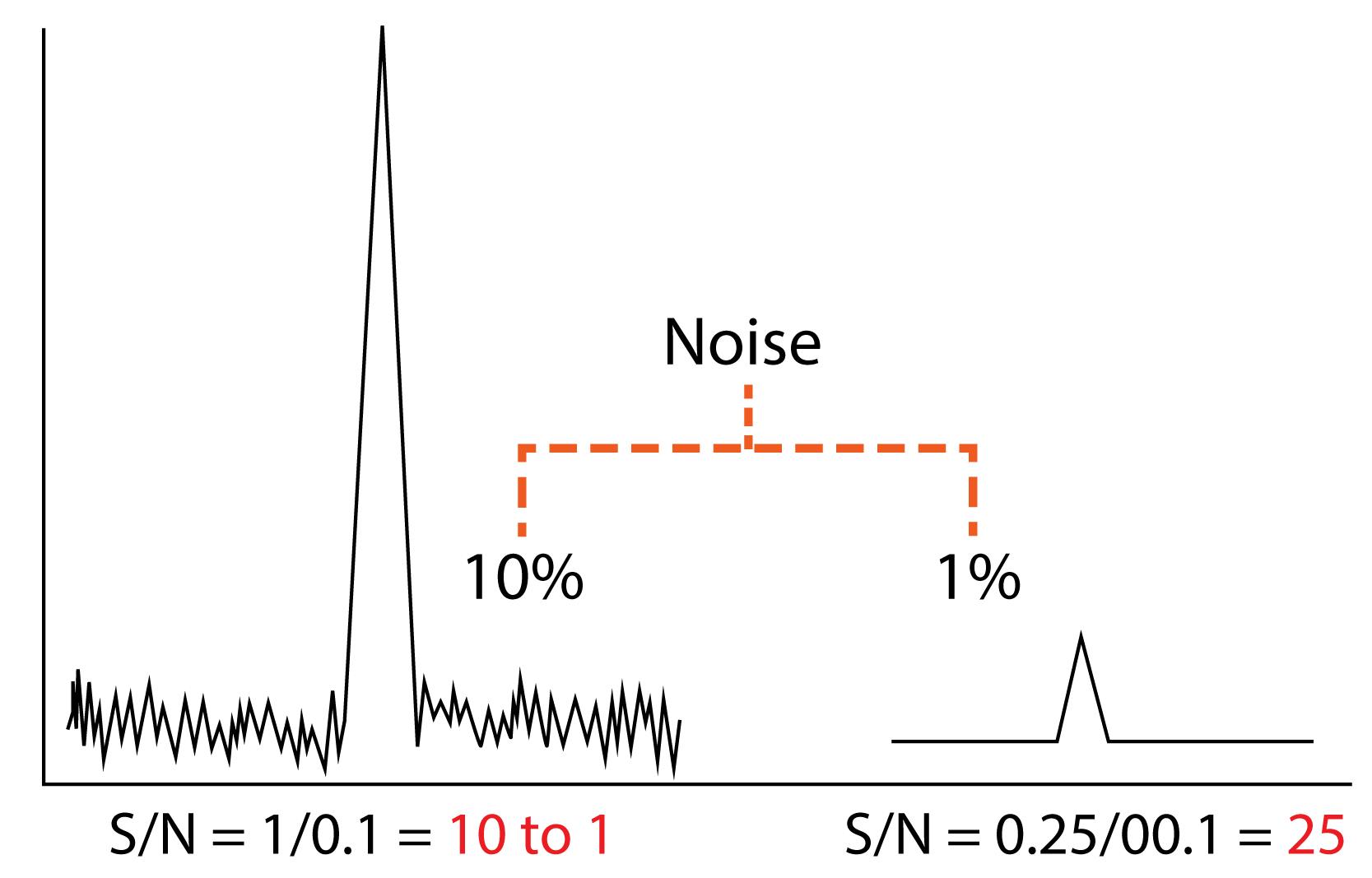
Sensitivity
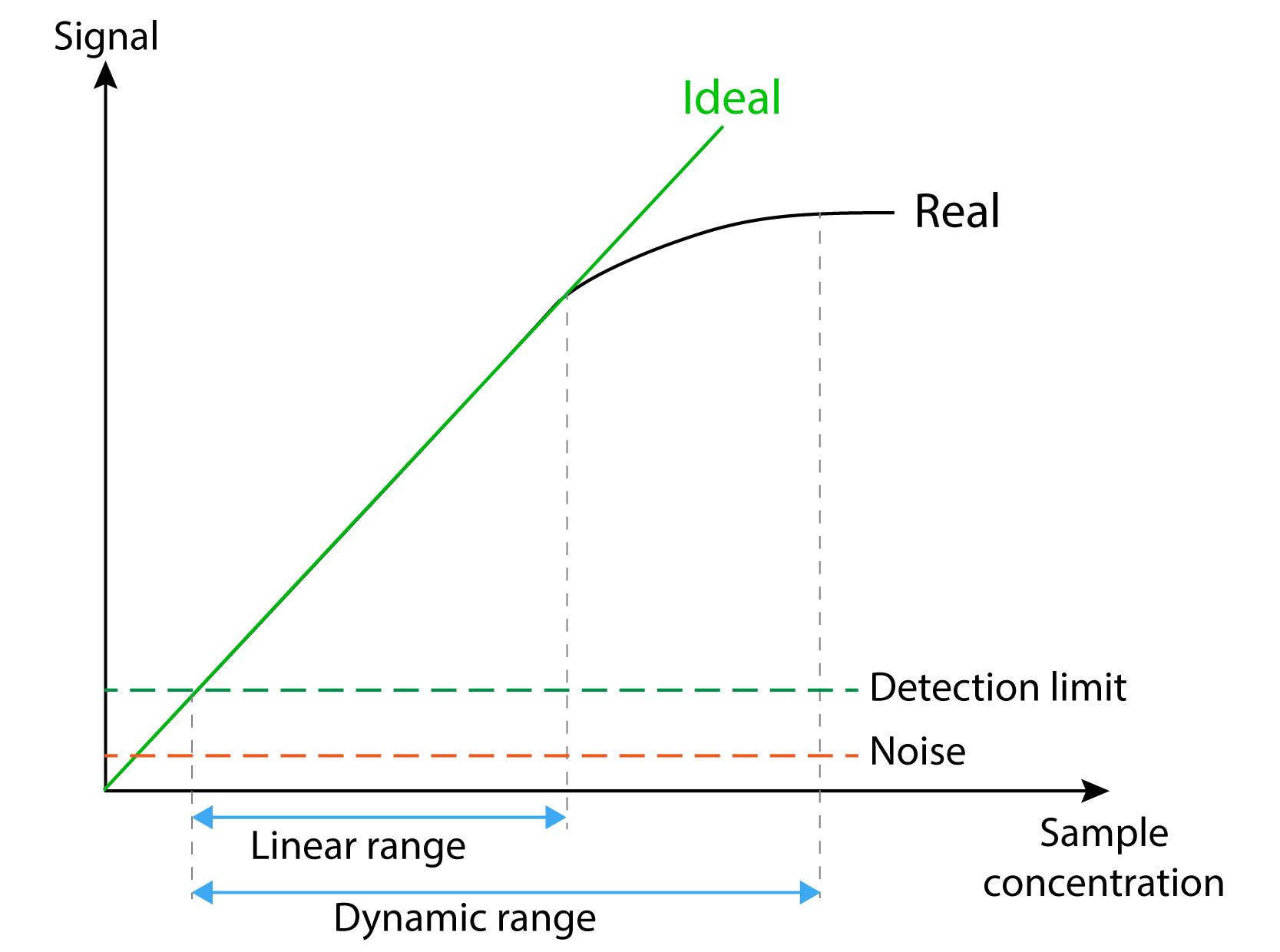
The detection limit of an individual analytical procedure is the lowest amount of analyte in a sample which can be detected but not necessarily quantitated as an exact value.’
When is a response considered significant? Methods have noise due to uncontrolled random errors in measurement and these can “drown out” low intensity responses. A method is considered sensitive if it can generate a precise and accurate response to our lowest desired concentration.
Linearity / Range
The linearity of an analytical procedure is its ability (within a given range) to obtain test result which are directly proportional to the concentration (amount) of analyte in the sample
‘The range of an analytical procedure is the interval between the upper and lower concentration (amounts) of analyte in the sample (including these concentrations) for which it has been demonstrated that the analytical procedure has a suitable level of precision, accuracy and linearity
linearity is where we obtain accurate results over a specified range of concentrations. Linearity and range are interlinked, if we don’t assess over a range then we just get accuracy. This shows that we have an acceptable correlation between concentration and response
Robustness
‘The robustness of an analytical procedure is a measure of its capacity to remain unaffected by small, but deliberate variations in method parameters and provides an indication of its reliability during normal usage.’
Robustness is where we show that the method is capable of coping with variability on key method parameters (such as pH, mobile phase composition, columns etc.). We do this by deliberately varying (bracketing) parameters around the specified value and assessing how much this affects method performance. Robustness is typically late in validation, so if there are any issues it’s a big deal to rectify them however in a QBD approach, we vary key parameters during method development to alert us early to any issues so that we can develop them out.
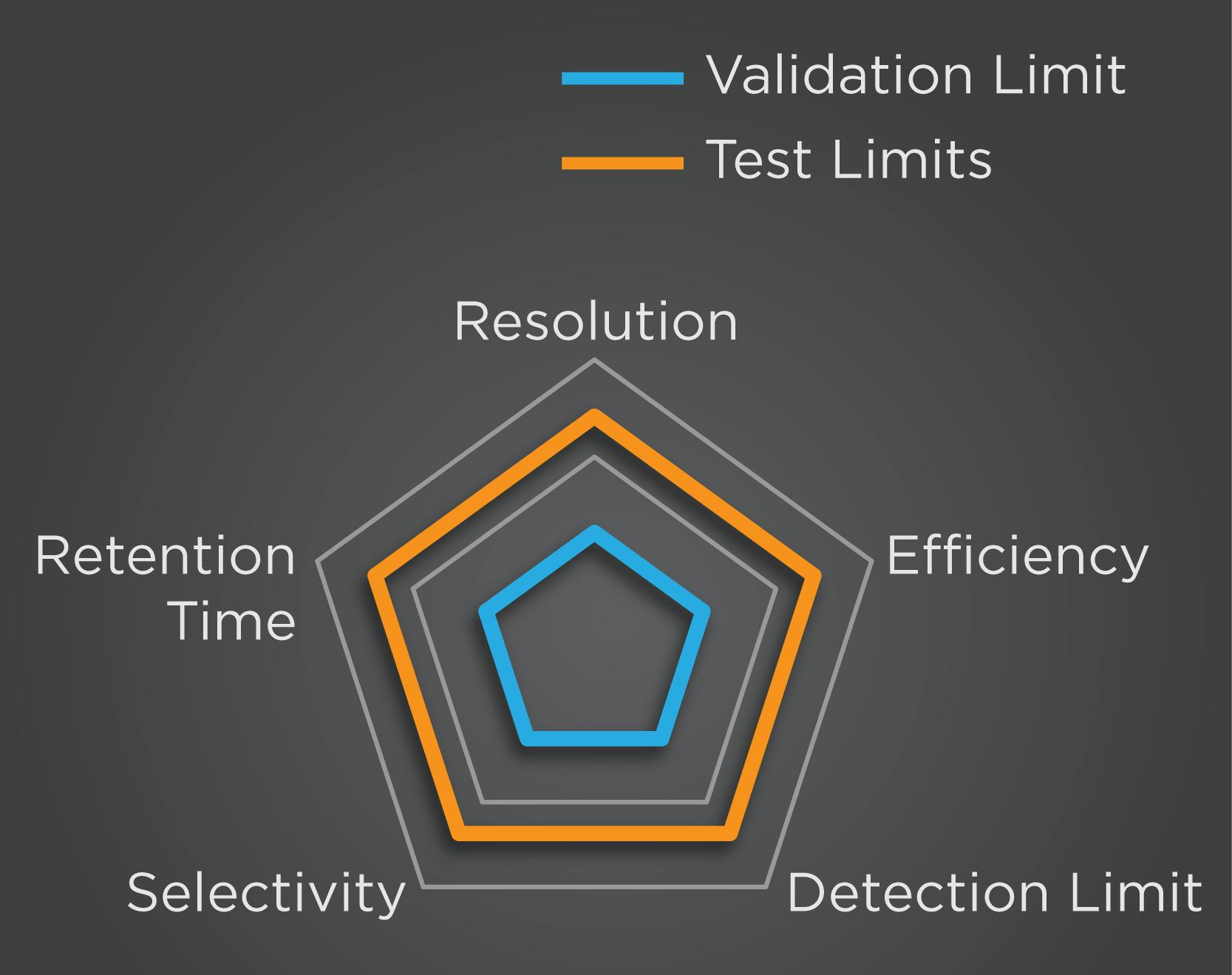
What do I need to do to validate my method?
Validating a method can be done with as little as 9 standards (and a matrix blank) if they are chose properly, with 3 at a low level, 3 at a mid level and 3 at a high level.
Specificity: The matrix blank should have all the same components as the sample just without the target. If we see no signal in the blank but signal in our sample, then we have a specific method
Accuracy: The standards are of a known concentration so we can measure how close the response is from the true response to give an understanding of the accuracy of each standard
Precision: If we have three points at each level of the curve, we can use them as replicates in order to measure precision at the low, mid and high levels
Sensitivity: The three points at the low level can be measured for their signal to noise ratio to ensure this is above a critical value
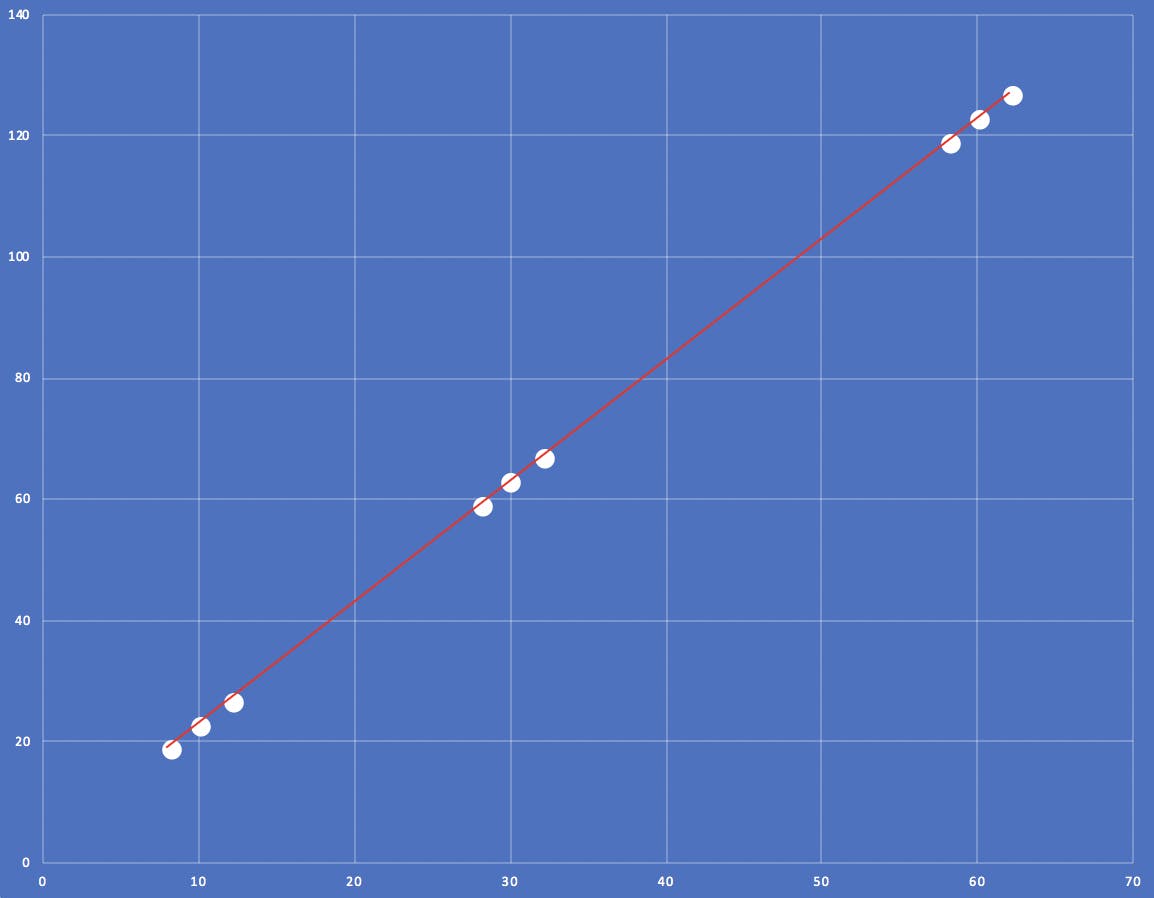
Linearity/Range: Having three levels across the concentration range allows us to establish a relationship across it and apply a linear regression model. The lower and higher levels can demonstrate the range that the method is capable of.
Robustness: Analysis of key variables should be carried out beforehand and small changes of these can be done and then the standards are run again to see if there was any effect
Validation should be formality - purely an exercise in obtaining a set of formal data, there should be no surprises, we should have already shown the method capabilities long before validation – during development and pre-validation.
To understand further how we test for these aspects, please see our training course Analytical Method Validation