22 Jan 2020
Chromatography Column Health Check

These are a small example of the type of comments made in labs all the time, especially by “lab hoarders” who keep every piece of equipment they have and take in every free piece that was destined for disposal, regardless of working condition. Having supplies around can sometimes be helpful but clutter means you lose control of knowing what is good in the lab and what is waste.
As the Christmas decorations come down, there is the opportunity to have an early spring clean to get the new year off to the best start. What about that drawer full of HPLC columns that have been used but replaced by newer, smaller, faster columns? Columns that previous lab members used but left behind when they moved on. Are they worth keeping or are they now metal tubes with not-so-magic sand in them?
A column health check can be useful to determine if a column has been stored correctly and if it is still capable of delivering the required chromatography and here some steps to do just that.
Visual inspection

By far the easiest and quickest test is to look at the column and the environment it has been stored in. Have both ends been capped to prevent the stationary phase from drying out? Has it been stored next to a heat source or exposed to other potentially damaging elements? Is there an associated logbook of the age and use of the column?
Pressure Test

Measuring pressure on a chromatography system is akin to an x-ray in that it allows you to look inside your instrument without having to open it up. Connect the column and begin at a low flow rate to not shock the column, pressure is directly proportional to viscosity (η), column length (L), and flow rate (F) so a lower flow will result in a lower initial pressure on the column. Slowly ramp up to the normal operating flow and monitor (record if possible) the pressure and compare this to previously recorded pressures if available (compare to column guide if not). Low pressure, high pressure and unsteady pressure are indications of column issues and they need to be investigated before the column can be used.
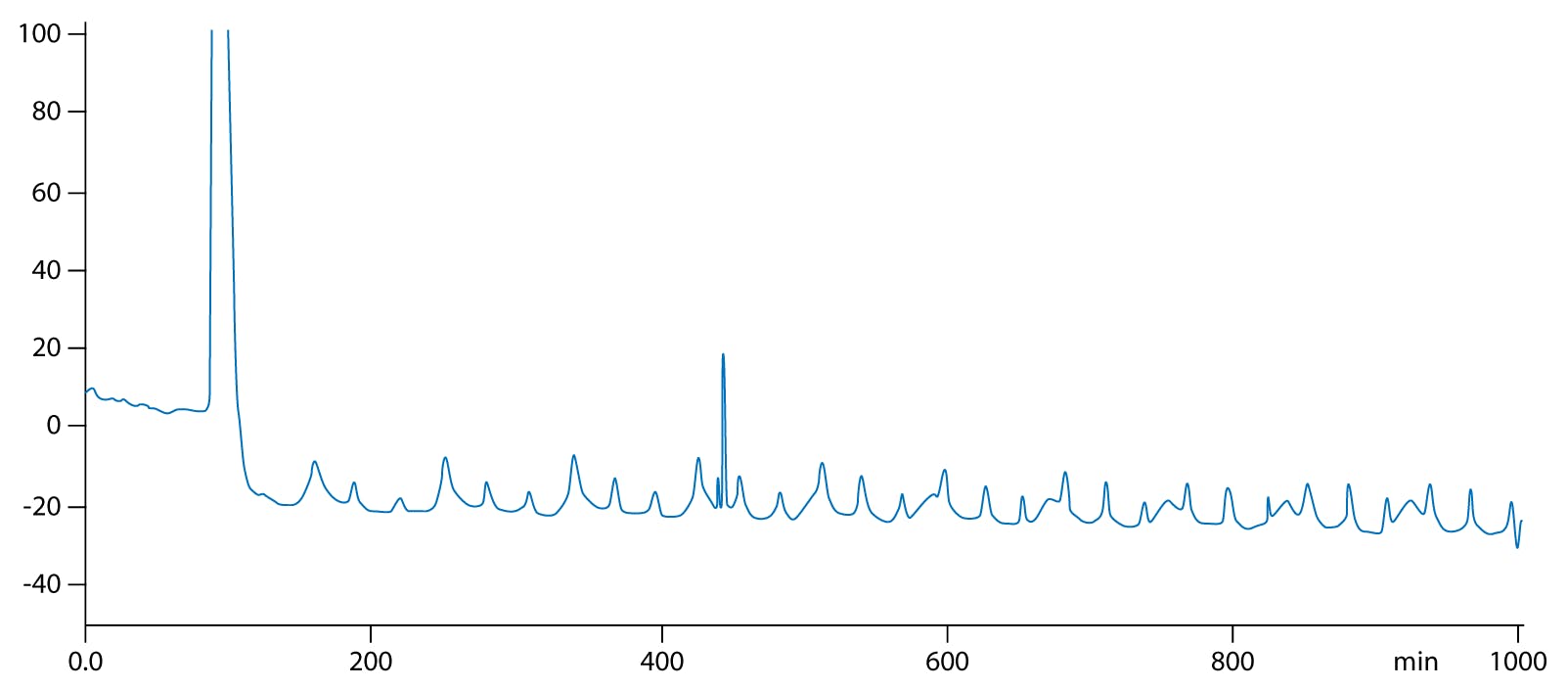
Figure 1: Observing ripples in a baseline acquisition.
Conditioning
Each column comes with a several documents which I’m sure every analyst has read in great detail and in them it will have the conditioning conditions for the column to ensure it is in the correct state for use, whether this is wetting, washing or correcting phase collapse. Performing these steps will improve the repeatability of the tests to be performed on the column.
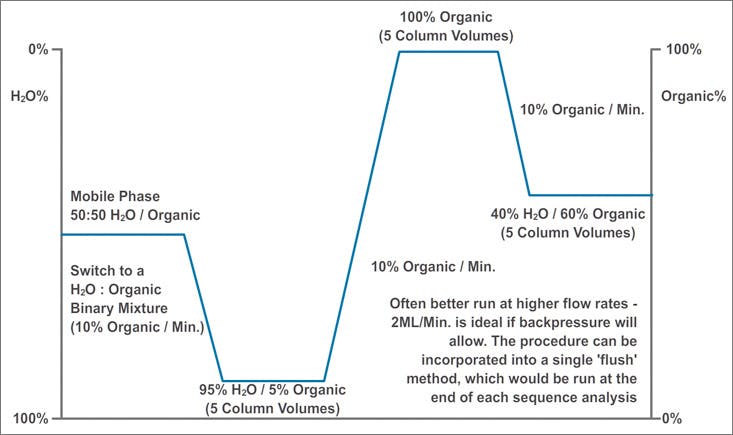
Figure 2: An example of conditioning for a RP HPLC column.
Run a test mix
A baseline is required to determine if a column is still capable of the separation required of it and that can be from a chromatogram from an earlier run or once again going back to that useful information that comes with every column. Test mixes can be purchased for any column chemistry, but they can also be put together with compounds you have inside the lab if they are testing the column in similar ways to the analysis required.
Look for the key parameters:
| Capacity factor – Is the column still able to retain the compounds? | |
| Selectivity – Is the column still able to separate the compounds of interest? | |
| Efficiency – Is the column still able to produce sharp peaks (a great indicator of column life) | |
| Resolution – Does the column meet the minimum requirements of the method for the critical pairs? |
Also look at peak shape to help identify column issues, tailing and front are very valuable for diagnosing issues in columns and systems.
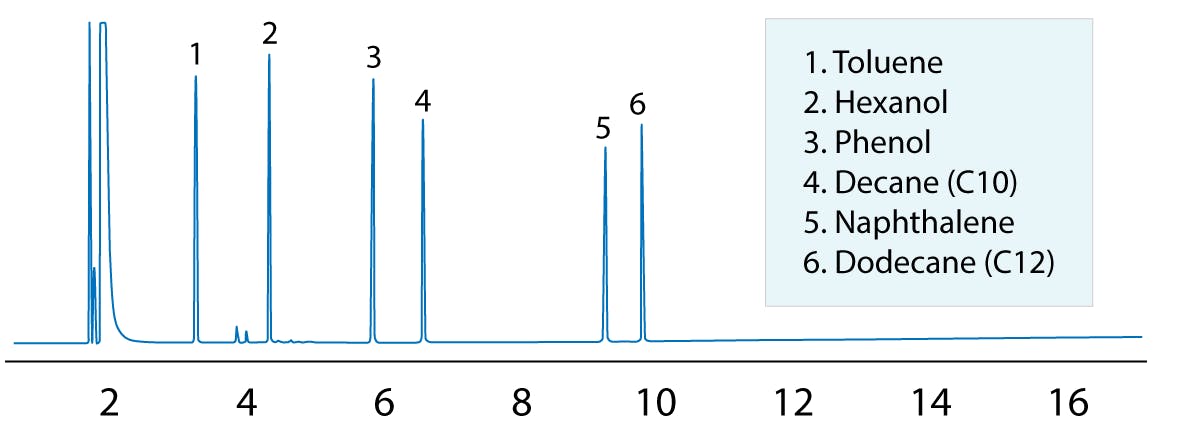
Figure 3: Test mix for GC Columns.
These checks could be performed on a routine basis to make sure the column is still acceptable for the method but also as an early warning system for maintenance or replacement. If the column fails any of these tests, it doesn’t necessarily mean that the column can be thrown away as there are remedy steps that can be taken to bring it back to working condition, for example an organic solvent backflush on certain HPLC columns. But always check the documentation that comes with the column to check if the column flow can be reversed and which conditions are suitable.