28 Aug 2017
Hydrophilic Interaction Chromatography - Practical Insights
I’ve spoken to a lot of people who recall their experience with HILIC along the lines of –‘oh we tried HILIC and it didn’t work for us’ or ‘we took our reversed phase gradient and ran it backwards’.
Hydrophilic Interaction Chromatography can be a very useful tool in our analytical armoury, however there are some practical peculiarities to this chromatographic mode which need to be understood in order to ensure success.
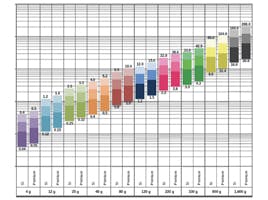
Equilibration tends to be the downfall of many people trying HILIC for the first time. One of the important mechanisms in HILIC chromatography is the partition of analytes between the bulk eluent and a water enriched layer at the stationary phase surface. This layer needs to time to form and equilibrate and as such a ‘new’ column will typically need to be equilibrated with 50 – 100 column volumes prior to use and re-equilibration between injections in gradient HILIC mode will require between 10 and 20 column volumes to obtain reproducible chromatography in our experience. To calculate column volume;
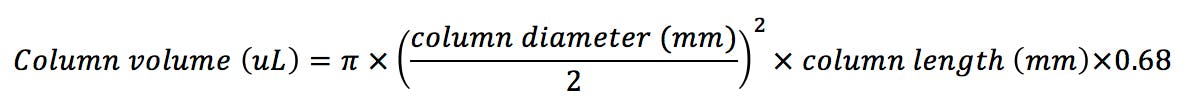
The nature of the ‘adsorbed’ water enriched layer dictates that the eluent must contain at least 3% aqueous by volume and our experience is that sticking to an upper limit of 95% organic helps with practical reproducibility.
Further, below around 60% organic (the actual lower limit will vary depending upon the nature of the stationary phase and eluent) one enters a more typical reversed phase condition and again reproducibility may be affected and the results obtained more difficult to interpret. The re-equilibration of the column takes longer if the upper limit of the gradient exceeds 50% aqueous.
Acetonitrile isn’t the only organic! Propanol is a weaker solvent than Acetonitrile and Methanol is eluotropically stronger. Adding 5% of either (90:5:5 Acetonitrile:IPA:Water for example) of these can really help to ‘finesse’ a separation.
When attempting HILIC separations for the first time, try using a diol phase. These cannot be ionised within our usual pH working range and hence the mechanism of the separation is more easily understood as there are no electrostatic interactions between the analyte and the bonded phase.
The pH of the eluent can have a powerful effect on selectivity and retention, but one needs to properly understand the mechanisms. Amide phases are cationic under more acidic conditions, which can reduce the retention of bases and promote the retention of acids due to electrostatic repulsion or attraction. Bare silica phases become increasingly anionic as we raise the eluent pH above 5, so reducing the retention of acids and increasing the retention of bases.
It is therefore important to understand the pKa of both the stationary phase and analyte in order to understand chromatographic behaviour under varying pH conditions.
Varying the degree of analyte ionisation can promote retention in HILIC mode, even when using diol stationary phases. Simply put, ionised analytes are more polar and will therefore partition more strongly into the polar aqueous layer adsorbed at the stationary phase surface.
The pH of the solvent system will vary when aqueous eluents are combined with higher amounts of organic. Expect a shift of between 1 and 1.5pH units towards neutral depending upon the amount of organic solvent in the eluent. This is particularly important when considering the ionisation state of either the analyte or the stationary phase.
Buffers can also have a pronounced effect on retention, and often on selectivity in HILIC mode and a sufficiently high buffer strength is often essential to maintain good peak shape and reproducible retention times. The nature of the buffer used and its concentration are primary method development parameters. The buffer type and strength will regulate the polarity of the adsorbed water layer, act as a counter ion for any ionised stationary phase or analyte moieties and will aid with the regulation of ionised sites when electrostatic retention mechanisms are in play. Ammonium formate and acetate buffers are popular in HILIC mode and a useful approach is to alter the ionic strength from 5mM to 20mM to discover the effect on both retention and selectivity.
It is very important to maintain a constant ionic strength when running gradients in HILIC mode. To aid in this approach it is useful to use mixed mobile phases, for example, mobile phase A is made up of 100% aqueous buffer and mobile phase B should contain 95% acetonitrile and 5% aqueous buffer; both mobile phases could be prepared by adding 5% of concentrated aqueous buffer to either water or acetonitrile. This approach helps to mitigate against changes in the retention mechanisms in play during the gradient which can lead to irreproducibility.
HILIC separations are particularly susceptible to differences between the eluotropic strength of the sample diluent and the eluent when the sample is injected. Broad, tailing or split peaks can all result from a mismatch in eluotropic strength between the sample diluent and the eluent. One should strive wherever possible to inject the sample using a diluent which is either equal to or less powerfully eluting than the initial eluent composition and in practical terms the sample diluent should contain no more than 25% water by volume. Altering the diluent pH or ionic strength can often help to overcome solubility issues.
The HILIC mechanism is so susceptible to these changes that one should also consider the nature of the syringe or needle wash solvent, maintaining as high a percentage of organic solvent as possible.
A great way to investigate HILIC separations is to run a 5 – 40% aqueous gradient using a diol type phase with 10mM buffer (constant ionic strength) at pH 2.5, 4 and 6 and assess the results obtained for any adjustments to pH or buffer strength that might be required to obtained suitable retention selectivity or resolution. If there are many critical peak pairs, it is more effective to change to a different stationary phase such as an amide or bare silica phase, however note that careful thought should be given to the various ionisation state of analyte and stationary phase surface when interpreting results with these phases as outlined above.