19 Mar 2019
Polymeric HPLC Columns
New Distributors for Shodex
Element became a distributor of Shodex HPLC columns at the beginning of 2019. With this new addition we complement our vast range of products with a complete family of polymer-based columns, designed for reversed phase, HILIC, ion chromatography, size exclusion, and more.
Shodex is the chromatography brand of Showa Denko K.K., a Japanese group of companies that began its history in the 1930s manufacturing fertilisers. Today, they manufacture products as varied as industrial chemicals and gases, ceramic materials, electronics, or aluminium. Your favourite soft drink could very well come in a Showa Denko can. In the 1960s Showa Denko started to produce synthetic polymers, and their quality control lab made their own size exclusion columns. That was not strange at the time: there were no commercial column suppliers, so every lab would produce and pack their own. But the Showa people perfected their art to the point that, in 1974, they decided their columns were good enough to sell, and Shodex was born.
Silica Benefits and Limitations
The most common solid support currently used in analytical liquid chromatography is, without a doubt, silica. Through a sol-gel condensation, precursor molecules such as TEOS (tetraethoxysilane) are condensed and cured to produce microscopic spherical silica particles. Among the interesting properties of this material are mechanical strength, controlled pore size and volume, and high surface areas. But the real key to the success of silica particles has been the ability to modify their surface chemistry: through a process called functionalisation, a variety of organic stationary phases can be grafted on the particles, resulting in a truly versatile range of columns.
Silica supports, however, have some limitations. The stationary phase cannot cover a hundred per cent of the silica surface, and a number of silanol groups remain exposed. These highly reactive groups can interact with the sample being analysed, resulting in distorted chromatographic peaks, low recoveries due to sample adsorption, shifting retention times and, in some cases, chemical degradation of the sample.
On the other hand, the stationary phase itself is liable to degradation. With continued use, the ligands are hydrolysed and eluted by the mobile phase, resulting in what is known as column bleed. The loss of stationary phase brings a loss of retention and loading capacity, as well as more silanol groups being exposed. Also, some detectors can respond to column bleed, resulting in a gradual distortion of the baseline, increased noise, reduced sensitivity, etc. All these effects combined eventually result in column failure.
Two further weaknesses are associated with silica-based chromatography. The first one affects hydrophobic stationary phases, such as the widely used C18 or C8 chemistries. If exposed to highly aqueous mobile phases, these phases “retreat” away from contact with the eluent, in a process called dewetting or, colloquially, “phase collapse”. The second weakness, perhaps the most restrictive, is that silica is only compatible with a relatively narrow pH range, traditionally between 2 and 8. Very acidic eluents accelerate the hydrolysis of the stationary phase and column performance decays very quickly. At basic pH, on the other hand, silica itself becomes soluble and the particle disintegrates into sub-micron fragments, sometimes called fines. Fines block the outlet frit of the column, resulting in increased pressures, and have a negative effect on the performance and robustness of evaporative detectors like ELSD, CAD, or MS.
Great improvements have been introduced in the synthesis and derivatisation of silica particles to mitigate these drawbacks, and this continues to be a very exciting area of research. But since the beginning of chromatography there has been an interest in experimenting with alternative support materials that may be unaffected by these limitations.
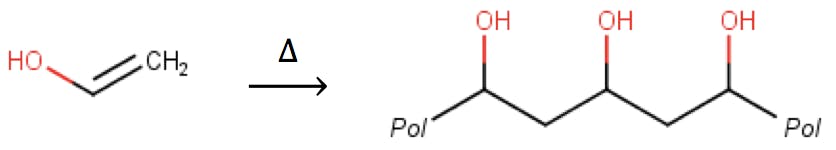
One of the best-known advantages of polymer resins is their excellent chemical stability under a wide range of pH, in many cases as low as 1 and as alkaline as 13. Thanks to this stability, polymers are the preferred support for ion exchange chromatography and other techniques that require very acidic or very basic eluents. Due to their resistance to hydrolysis, polymers also tend to suffer less bleed than silica particles, resulting in cleaner baselines, consistent retention times, and low operating pressures. All these desirable properties often result in greater column durability. In some cases, polymeric columns have been shown to last two to three times longer than silica columns under equivalent conditions.
I’s not all advantages, however: polymers do not have the mechanical strength of silica particles, and in general cannot withstand very high pressures. Another notorious limitation of some polymeric particles is their tendency to “swell” in some organic solvents. These weaknesses can be improved by polymerising the base material with a carefully chosen co-monomer, in a process called “cross-linking”.
PS-DVB Particles
Resins based on polystyrene cross-linked with divinylbenzene (PS-DVB) are some of the best studied polymeric substrates, and the most widely used in ion chromatography. Resins made of pure polystyrene are soft and readily soluble in organic solvent. But adding divinyl benzene increases the average molecular weight of the polymer chains, making them less soluble, and improves the mechanical properties of the resulting beads.
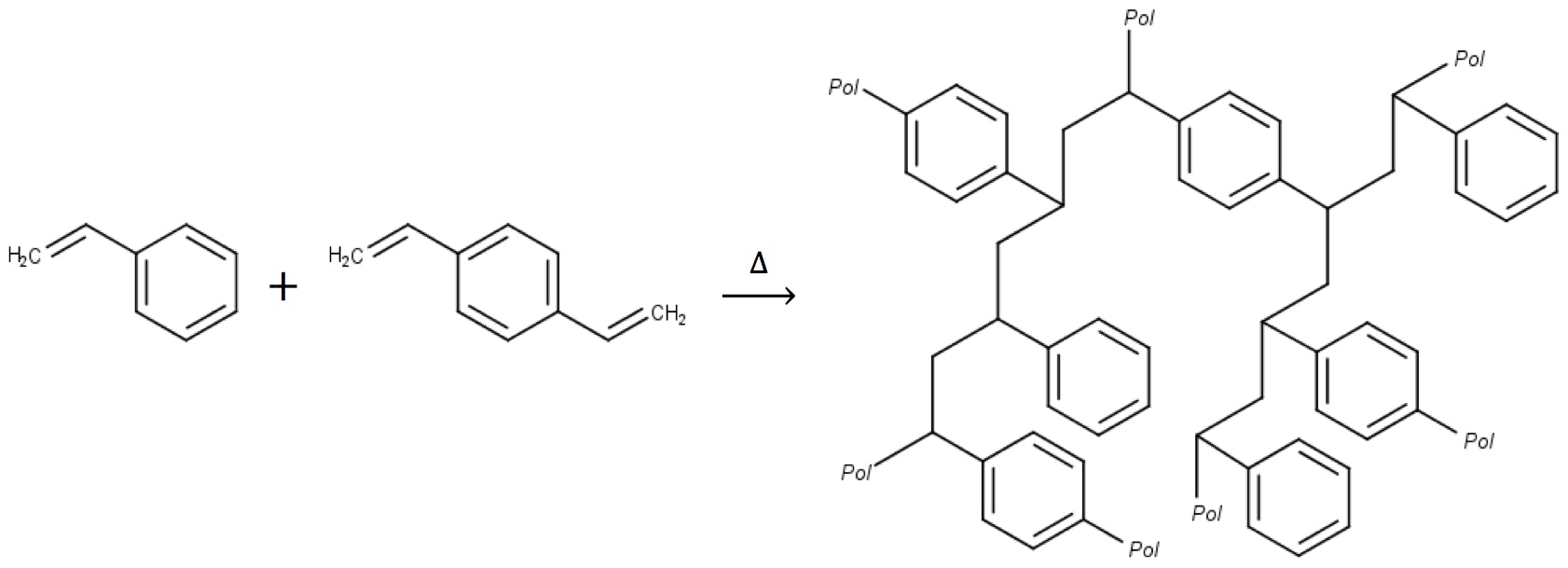
Traditionally, the resins have been classified as microporous (solid particles with small surface pores, a low surface area, and around 8% DVB cross-linking) and macroporous (particles with up to 500 Å pores, surface areas as high as 800 m2/g, and up to 55% DVB cross-linking). Both types are prepared by continuously stirring a suspension of monomer droplets in water. Adding an initiator such as benzoyl peroxide triggers the polymerisation and the polymer grows within the confines of the droplet. By regulating stirring speed and temperature, it is possible to control the size, and size distribution uniformity, of the resulting spherical beads. To synthesise porous resins, the droplets in suspension contain a diluent, chosen to be a good solvent for the monomers but not for the polymerised resin. Thus, as the polymer grows, nanometre-wide pools of the diluent become trapped within the matrix. After the polymerisation is complete the eluent is washed out, leaving a spongy but rigid structure.
Functionalisation of the Polymer
In broad terms, anion exchange chromatography is based on a positively charged stationary phase, which electrostatically attracts and retains the sample anions. Subsequently, anions in the eluent (typically carbonate or hydroxide) are attracted to the stationary phase and displace the sample anions, which gradually elute from the column.
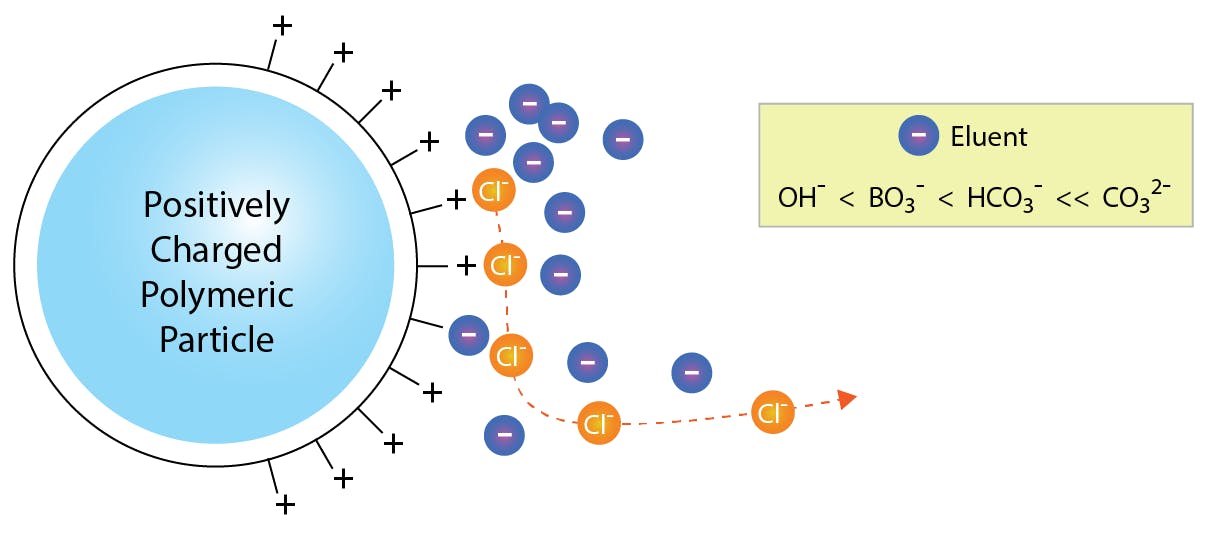
In cation exchange, the principle is inverted: negative charges on the stationary phase retain cations in the sample, and these are eluted by protons in the mobile phase.
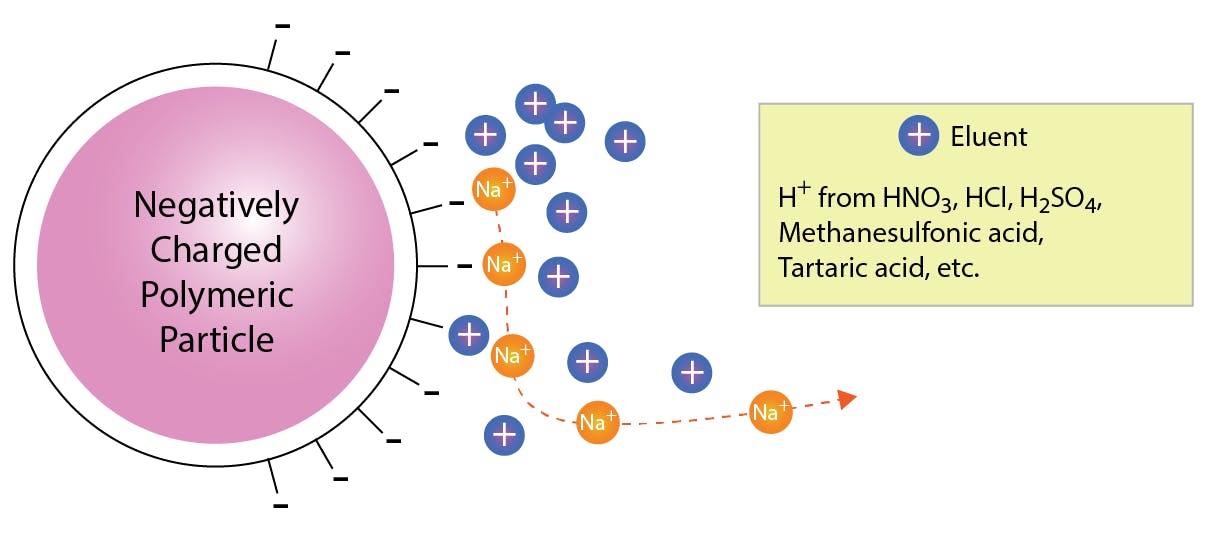
The PS-DVB resin is completely neutral, so it needs to be functionalised to enable these reactions. Fortunately, ion exchange sites and other functional substituents are readily introduced through a variety of reactions. A common functional group used in cation exchange is sulfonic acid, which is introduced onto the PS-DVB resin by treatment with concentrated sulfuric acid, or with chlorosulfonic acid followed by hydrolysis:
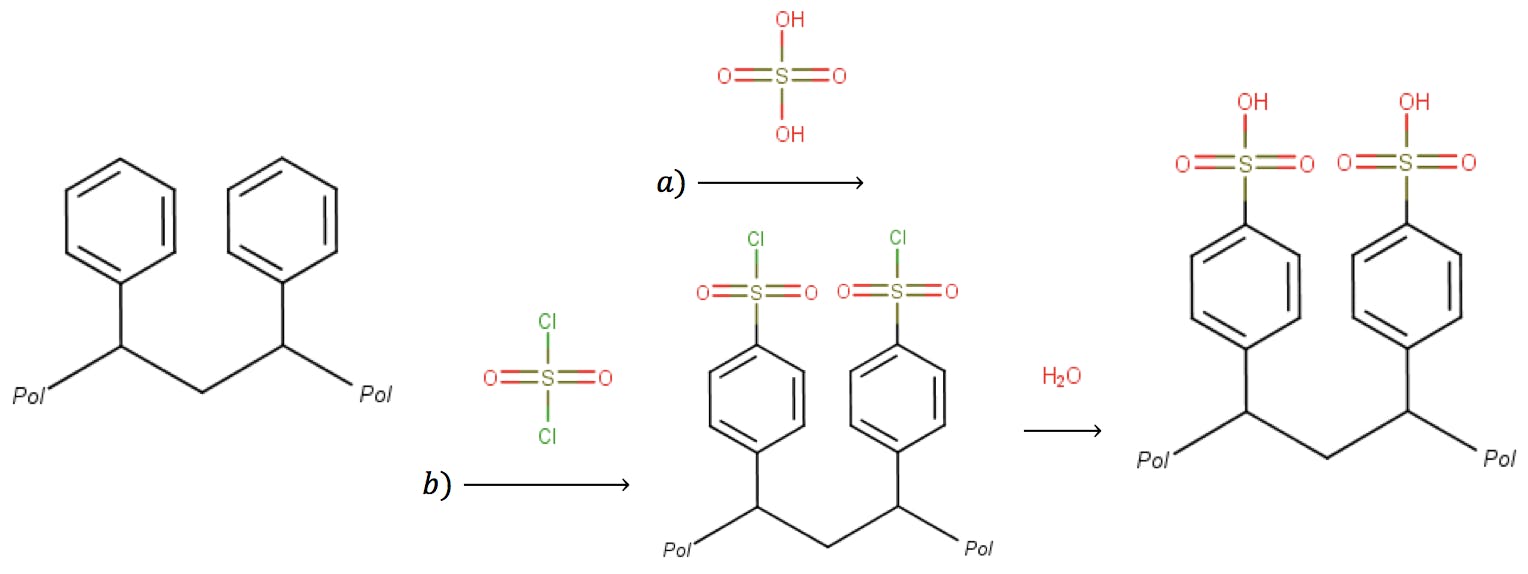
On porous resins, the sulfonation reaction proceeds on both the outer surface of the particles and in the large inner pore surface, easily grafting a high number of ion exchange sites. On microporous resins, however, the sulfonation can only reach the external surface of the particle. On these materials, the resin is first swollen by an organic solvent to allow the acid access to deeper phenyl moieties.
Anion exchange resins can be prepared by a two-step process. Initially, the benzene rings are derivatised via a Friedel-Crafts alkylation, introducing a chloromethyl group. In a second step, an amine (primary, secondary, or tertiary) reacts with the chloromethyl group to yield the final resin:
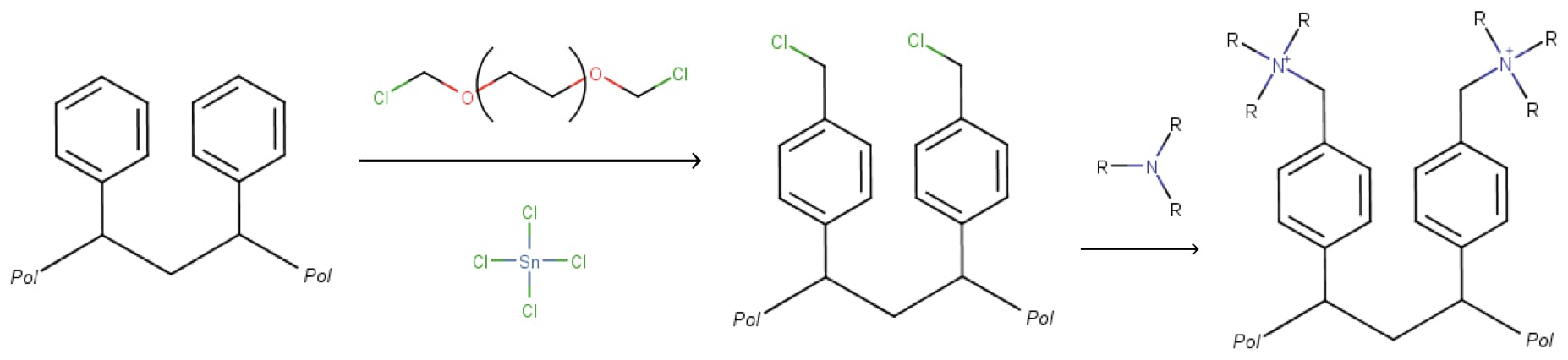
The number of sulfonated or aminated sites is important in ion chromatography, because it is directly related to the ability to retain and separate analyte species. This is commonly known as the resin “capacity”. Non-porous resins of very low capacities are inadequate for analytical chromatography because of their weak selectivity. On the other hand, very porous resins with high capacities tend to yield poor resolution because of diffusion and mass transfer band broadening effects. This is one of the reasons why ion chromatography particles have remained relatively large (up to 18μm are not unusual) whilst silica particles have reduced their sizes to less than 2μm in recent years: a delicate balance must be struck between particle size, surface area, porosity, mechanical strength, and ion exchange capacity.
Polymethacrylates
This balance is highly dependent on the material and the synthetic methods used to prepare the chromatographic media. One of the successful strategies followed by Shodex is to use a wide range of polymeric resins, functional groups, and ligands in the manufacture of their columns, combining the strengths of each material to best suit specific applications.
PS-DVB is extensively used in ligand exchange columns, where the requirement is to produce a strong sulfonated base material on which a metallic counter-ion is electrostatically immobilised. Columns in this range include the SUGAR SC, SP and KS series, or the RSpak DC-613, a mixed-mode support that combines ligand exchange and HILIC properties. PS-DVB is also the base material for an extensive range of GPC (gel permeation chromatography, or organic-based size exclusion) columns. In this application, the polymer is used without any surface modification: the desired characteristics are a highly rigid structure and precisely controlled pore sizes, which are obtained through extensive cross-linking. Shodex has gained a reputation for producing high quality, durable columns, with the GPC-K (optimised for chloroform as eluent), KF (for THF) and KD (for DMF as eluent) families. Each family of GPC columns is available in a carefully controlled range of pore sizes to analyse polymers of different molecular weights.
For ion exchange applications, where particle surface wettability, porosity, and ion exchange kinetics are important, Shodex has developed several columns based on polyhydroxymethacrylate resins:
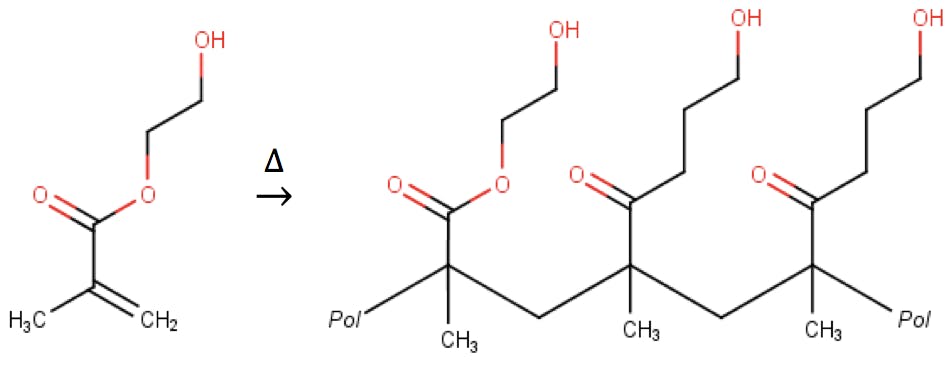
These base materials have excellent wettability characteristics in pure water eluents and buffers, but are also compatible with moderate amounts of methanol, acetonitrile, and other polar solvents. Typical pH ranges span from 2 to 12 or 13. Many of these particles are non-porous, to enhance separation kinetics, and their surfaces have been functionalised with sulfonyl, carboxylic acids, tertiary, or quaternary ammonium functional groups to deliver a complete range of ion separation solutions. Polyhydroxymethacrylates are the supports of choice for ion exchange (e.g. QA-825, DEAE-825, SP-825 columns), but also for GFC (aqueous size exclusion, for instance with the OHpak SB-800 HQ columns), affinity chromatography (AFpak APA-894, AFpak ACH-494) and other chromatographic modes.
Additionally, these materials exhibit low hydrophobic interactions with proteins, making them suitable for the separation of these compounds by ion exchange or size exclusion; as well as peptides, amino acids and nucleic acids.
Polyvinyl Alcohols
When the separation requires a particle that can withstand organic solvents, but is also compatible with pure water, polyvinyl alcohol (PVA) is an excellent resin. As it polymerises, PVA forms domains with a near-crystalline arrangement connected by amorphous chains, which gives the finished particles good mechanical strength and resistance to solvents.
As noted above, the synthetic flexibility of PVA even rivals that of silica particles. PVA resins can be functionalised with a wide range of ligands, ranging from hydrophobic (C18, C8) to polar (amino, diol) and cation exchangers (carboxylic acids). Because of the large number of hydroxylic groups on the surface of the particles, hydrophobic stationary phases do not suffer dewetting, even after being stored in pure water for extended periods. At the same time, the hydroxy groups provide enhanced retention of polar and very polar substances when compared to traditional ODS phases. Some columns, like the Asahipak ODP-50, are particularly effective at separating basic compounds without peak distortion, a challenging application for traditional silica-based ODS particles due to secondary interactions between the analyte and residual silanol groups.
This is the material of choice for polymer-based reversed phase (Asahipak range), hydrophilic interaction (HILICpak), or multi-solvent size exclusion columns (Asahipak GF-HQ, compatible with both aqueous and organic eluents). The HILICpak range is available with amino, quaternary ammonium, diol, and carboxylic functional groups in 5μm particle size and 100Å pores. Given the complex interactions between analyte, mobile phase, and stationary phase that underpin HILIC retention mechanisms, having a range of different chemistries is a powerful tool for method development. In all cases, the stationary phase exhibits very low bleed, making PVA-based columns an excellent match for mass spectrometry detectors.
More Polymer Columns from Element
The Shodex range is a valuable addition to the Crawford catalogue, but these are not our only polymeric columns. Agilent has a well-established range of polymeric columns designed for the separation of biomolecules. These include PLRP-S, originally developed by Polymer Laboratories, and introduced in 1984 for the separation of proteins by reversed phase; PL-SAX (Strong Anion Exchange) and PL-SCX (Strong Cation Exchange).
All these products are based on PS-DVB resins, stable across the whole pH range, and available in pore sizes from 100Å to 4000Å for the analysis of a wide range of biomolecules, from small peptides and oligonucleotides to large proteins and DNA fragments. An interesting feature of these products is that the resins are available in bulk quantities, seamlessly scaling up the analytical separation to preparative and process applications.
YMC offers polymeric columns, also targeting the analysis of biomolecules, in the YMC-BioPro range. These include the BioPro IEX columns, based on a polymethacrylate base material with anion and cation exchange functionalisation. The particles are available in porous (1000Å) and non-porous topologies and are stable between pH 2 and 12. The BioPro HIC column is based on 4μm, non-porous polymethacrylate particles, functionalised with butyl chains. They are stable at pH 2 to pH 12, and the material is compatible with moderate amounts (up to 50%) of methanol, ethanol, and other polar solvents.
Shodex columns will complement and enrich our current offer of polymeric columns for the analysis of biopolymers and small molecules. It is especially exciting to expand our HILIC method development kit with a few new phases. At the same time, we can now make available a complete range of high resolution ion chromatography and size exclusion columns, backed up by an extensive database of application methods and a long history of satisfied customers.