05 Aug 2019
Using Hydrogen Carrier Gas with Mass Spectrometric Detection
Our recent discussion on the use of hydrogen as a carrier for gas chromatography applications elicited many questions and comments. A particularly common question was ‘what are the considerations for using hydrogen carrier with MS detectors?’.
The answer – it’s possible but not straightforward!
There is a spectrum of issues when switching to hydrogen carrier but rarely have any of the issues been insurmountable with a bit of determination. Although the drivers for change may not be convincing at present, as the availability of helium decreases costs go up. This means that it’s time to get serious about making the change.
What follows is a brief discussion on the known issues and various approaches to adopting hydrogen as the carrier for GC-MS applications, to help inform decisions and practice.
Flow rates and column dimensions
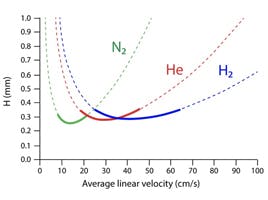
Hydrogen is around half as viscous as helium and there is a vacuum at the column outlet. This means that the head pressures required to generate the desired column flow rate can be too low for instrument pneumatic controls to deliver in a reproducible fashion. In most cases, the answer is to use a shorter, narrower internal diameter column, to achieve good linear velocity (>35 cm sec-1) at lower volumetric flow rates and head pressures that the instrument pneumatic systems can reproducibly deliver. Flow rates of between 0.3 and 0.4 mL/min are prevalent in literature, with linear velocity in the 40 cm sec-1 range or higher. This is appropriate as hydrogen is around twice as efficient as helium in this range.
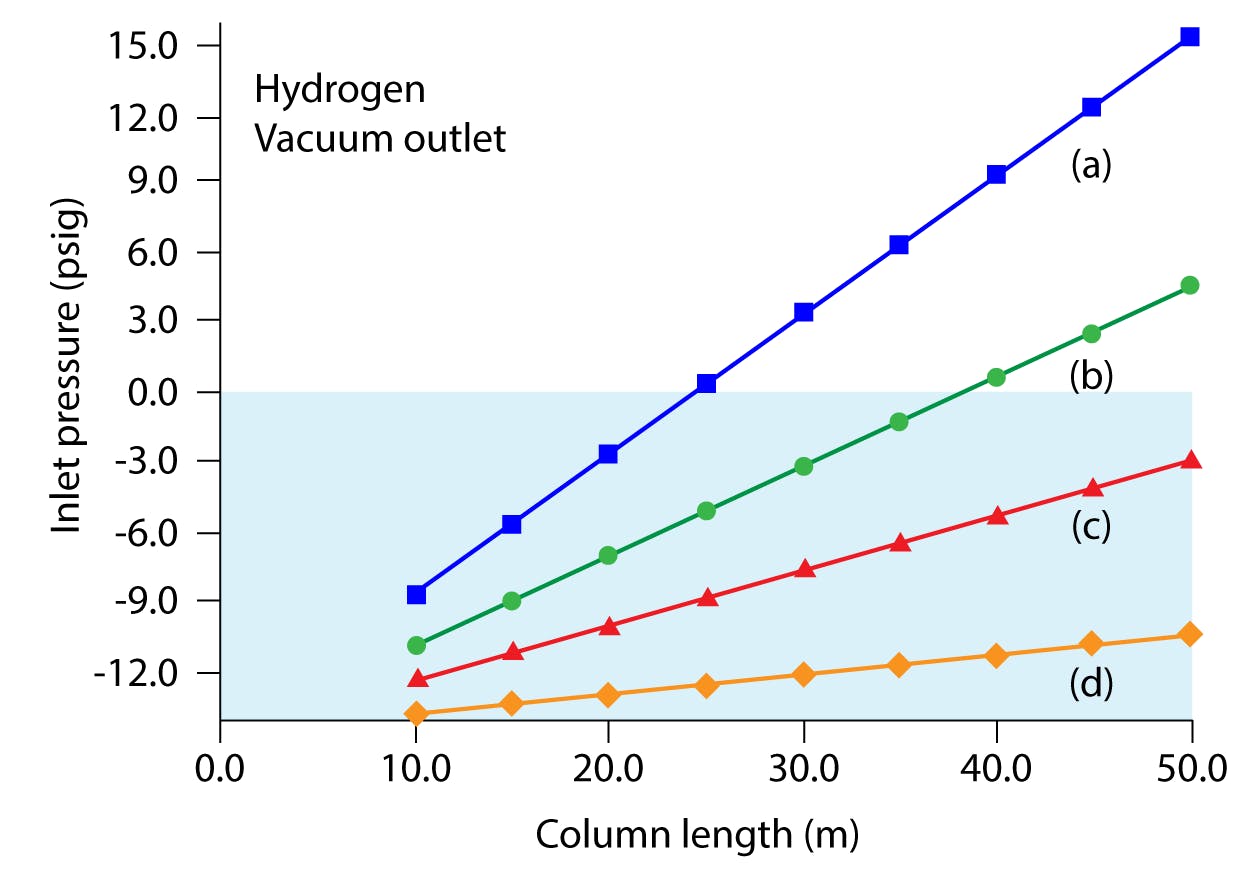
Figure 1: Plots of inlet pressure vs. column length, with hydrogen carrier gas and vacuum compensation on.
Column inner diameters (mm): (a) 0.20, (b) 0.25, (c) 0.32, (d) 0.53; column temperature: 50 °C; average linear velocity: 40 cm/s.
The blue shaded area designates negative inlet pressures.
Delivering lower volumes of hydrogen into the ion source offers other advantages as we will see later.
Two further considerations result from the use of narrow internal diameter columns;
1) The stationary phase capacity of the column will be more limited and therefore injection volumes or sample concentration may need to be reduced
2) Translation software may be required to maintain retention times between the original and new methods. As linear velocity changes the rate of change in temperature per unit time may need to be adjusted to maintain the elution order of the analytes. At the very least, verify the identity and elution order of analytes under the new column conditions.
A further impact of the use of lower head pressures is the rate at which the sample is transferred to the GC column, which can be further exacerbated using a lower viscosity carrier gas. To avoid issues of backflash (overfilling of the inlet liner due to solvent expansion, leading to carry-over and/or irreproducible peak areas), a pressure pulsed injection may be considered, which applies an increased head pressure to the system during the injection phase of analysis.
MS Vacuum System
Due to the lower viscosity of hydrogen, MS vacuum pumps must work harder to maintain the required vacuum levels. Some vacuum pumps are not capable of achieving the required vacuum.
Insufficient vacuum within the detector will result in lower inherent instrument sensitivity and may also risk a build-up of hydrogen within the detector. This is an obvious safety concern but will also increase the possibility of reactions between analyte ions and hydrogen carrier (more on this later). Monitor vacuum level and ensure the minimum vacuum level is being achieved (one manufacturer recommends a vacuum no lower than 5 x 10-5 torr for example).
More recently, instruments have been designed with pumping systems capable of achieving the required vacuum levels with hydrogen carrier. Check with the manufacturer before making the change to hydrogen. It is also important that the roughing pump outlet is directed into a vented fume-hood or outside.
MS Source Components
Most manufacturers will either offer a hydrogen specific ion source design or will provide change-out components to ensure that the source is optimally configured. In most cases, this will be componentry, ensuring the optimum ionisation efficiency within the ion source combined with ‘slotted’ components to allow the most effective pumping-away of the carrier gas from within the ion source.
There are also ad hoc reports of hydrogen carrier altering the metallurgic properties of certain metals used within ion sources, as well as changes to how magnets within the ion source, operate. Once again some of these components may need to be changed to work with hydrogen carrier. Contact the manufacturer for more advice on this subject.
Conditioning
Hydrogen is more likely than helium to displace contaminants which build up on roughened or unswept surfaces within the GC system and the mass spectrometer. For this reason, spectra acquired soon after changing the carrier gas will appear very noisy.
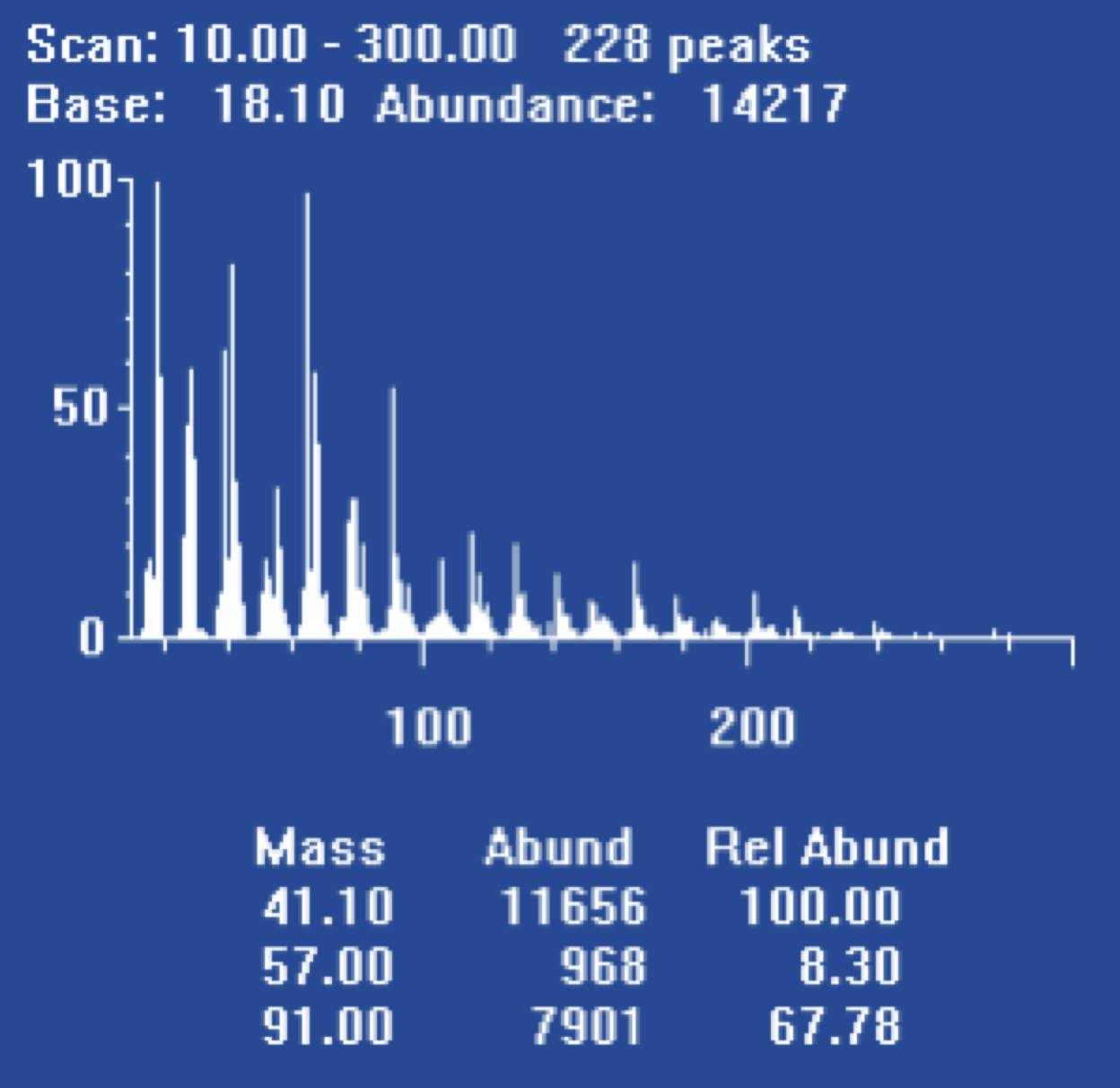
Figure 2: ‘Noisy’ spectrum obtained from PFTBA tune compound ‘scan’ on switching to hydrogen carrier gas.
Manufacturers suggest overnight ‘conditioning’ routines. These might include running at the highest source temperatures and reducing detector (electron multiplier) voltage with the filament turned on. Despite being in the early days of experimenting with hydrogen carrier for MS detection, conditioning periods of up to one week are common. Most manufacturers now suggest overnight conditioning routines.
Further, if the system is not properly conditioned, very pronounced chromatographic peak tailing may occur. In some cases, this tail may be chromatographic in origin (i.e. secondary interaction of the analyte with active sites within the system) but occasionally this may be due to the formation of analyte degradation products due to ion-molecule reactions with hydrogen in the ion source. This phenomenon will reduce significantly after proper conditioning.
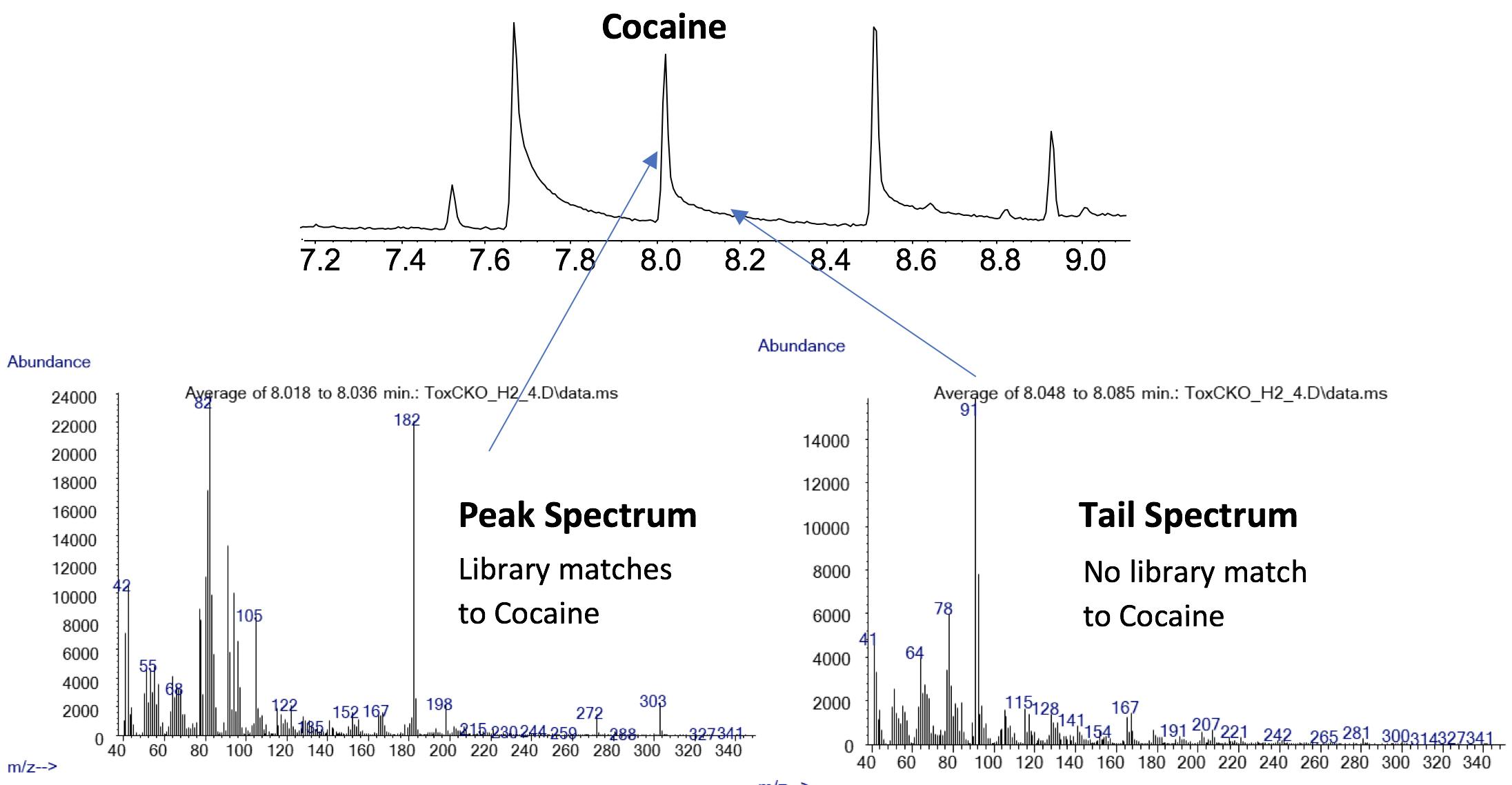
Figure 3: Spectra from a tailing GCMS peak which reveals that the tail contains a separate species – postulated to be a degradation product (Figure courtesy of Agilent Technologies, Santa Clara, USA)
The use of hydrogen does require more conditioning pre-analysis, even on standing overnight, but this does not significantly impact on the ‘practical usability’ of hydrogen as a carrier.
Sensitivity
Typically, a factor of between 2 - 5 fold reduction in sensitivity is to be expected when using hydrogen as the carrier gas. Baselines may be noisier; however, this can be offset by improvements in peak efficiency due to the higher inherent efficiency of hydrogen carrier and the (typically) shorter retention times.
The reduction in sensitivity is typically attributed to the higher source pressures (lower vacuum) associated with the decreased pumping efficiency that comes from using hydrogen. Therefore, using the optimum volumetric flow into the ion source, hydrogen approved ion source components, and highly efficient pumping systems is a pre-requisite. Especially, when carrying out trace analysis.
Ion-molecule reactions
Hydrogen is inherently more reactive than helium. It is possible, especially at elevated source pressures, to induce chemical ionisation (CI) ‘like’ reactions between analytes and hydrogen ions within the source to form an M+1 ion. Some reports cite hydrogen-induced degradation, in-source reactions, such as the reduction of nitrobenzene to aniline, or hydrogenation of unsaturated species. However, the literature on ion-molecule reactions in electron ionisation (EI) GC-MS, using hydrogen carrier, does not reveal a significant amount of supporting information.
Only one or two real examples of changes in analyte composition which can be attributed to hydrogen ion-molecule reactions are known. The general advice would be to assess the possibility of ion-molecule reactions when translating an established method or developing new ones.
When using chlorinated solvents such a dichloromethane or hydrogen sulphide (H2S), encountering the formation of hydrochloric acid at the elevated temperatures within the GC inlet is possible. Reactions in the MS ion source may also be encountered if the temperature is high enough. This acid has the potential to strip deactivation away from the liner and GC column, leading to tailing peaks and/or problems with quantitative reproducibility or linearity. For this reason, working with these solvents when using hydrogen as the carrier is to be avoided. Consider using a highly deactivated liner with bottom restriction, to avoid contact of the analyte and solvent with the hot metal surfaces at the bottom of the inlet. Changing GC consumables regularly is also advisable.
MS Spectra and Tuning
Most MS spectra remain very similar. However, there is a tendency for the quality of spectral ‘match’ to drop when using hydrogen carrier gas. Typically, this doesn’t affect positive identification. It's rare to experience a situation where qualifier ion ratios need to be adjusted when quantifying in selected ion mode; however, these limitations should be acknowledged and considered when converting.
Although not all spectra are affected in the same way, there are examples where appreciable changes occur in certain ion ratios between spectra obtained in either helium or hydrogen carrier gas. There are also examples where extra peaks are seen (usually at low abundance) in the spectra acquired with hydrogen as the carrier.
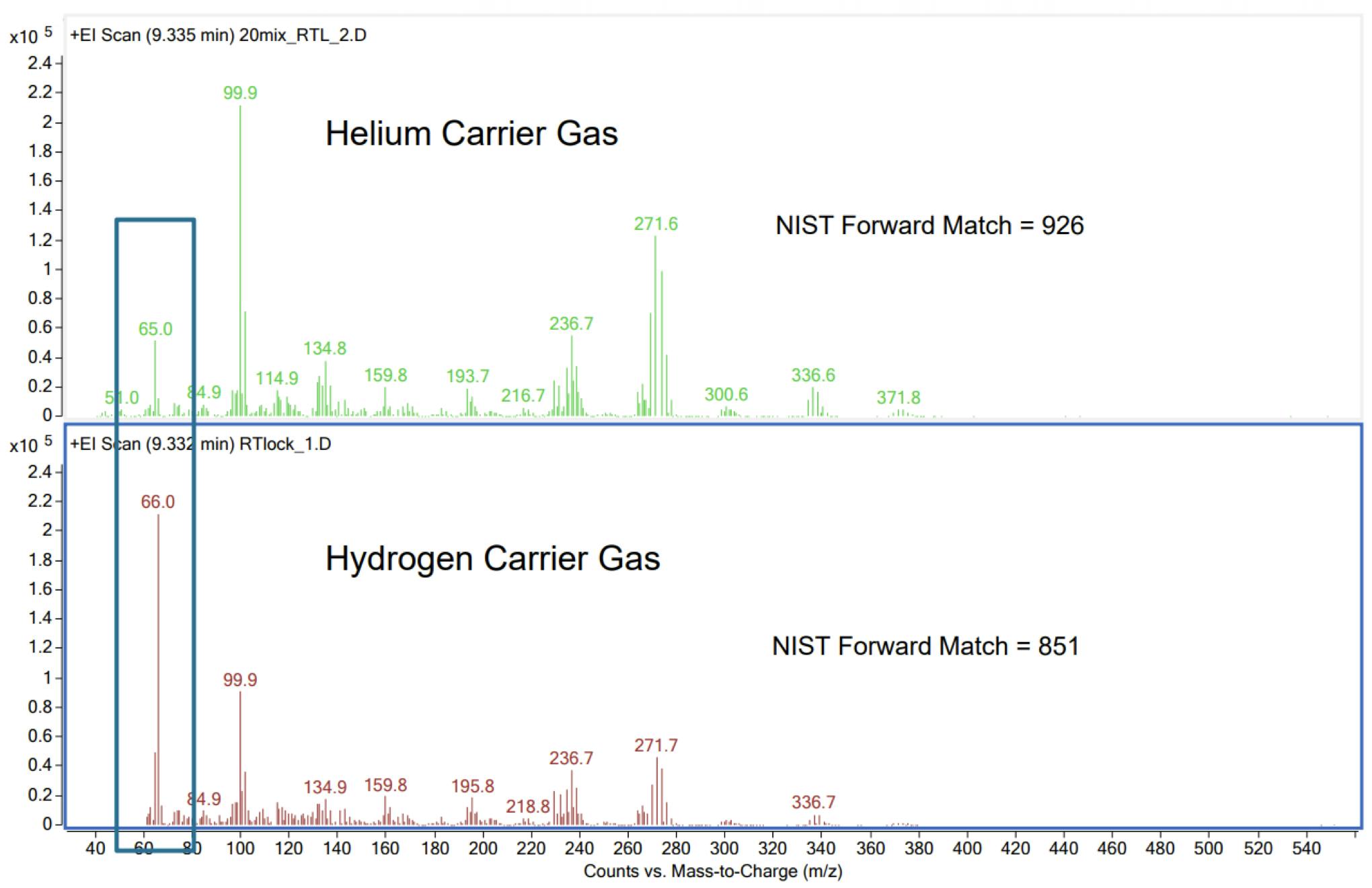
Figure 4: Spectral differences for EI-GCMS analysis of Lindane (Figure courtesy of Agilent Technologies, Santa Clara, USA)
In terms of tuning, in our experience, electronic ionisation tuning characteristics obtained using perfluorotributylamine (PFTBA, HEPTACOSA) are usually consistent with those obtained using helium carrier, and in most instances, a satisfactory tune can be obtained (i.e. the tune ‘passes’ and is within the tolerance of the instrument manufacturers tune targets).
Nevertheless, there are regulatory methods which use alternative tune compounds to PFTBA and also require certain relative ion abundances to be within defined tolerances. Decafluorotriphenylphosphine (DFTPP) for EPA method 8270 appears to give reasonable tune agreement between the two carrier gases. However, it is reported that bromofluorobenzene (BFB), used for EPA methods 524, 624 and 8260B, for example, can suffer from skewing of some key ion ratios when using hydrogen carrier. Especially, in qualifying the relationship between masses 95 and 96 m/z.
Possible. But not straightforward
In summary, it is possible to change carrier gas to hydrogen. But not straightforward.
Major considerations for converting to hydrogen carrier gas when using MS detectors are;
- Use of reduced dimension columns at higher linear velocity but lower volumetric flow
- Use of efficient vacuum pumps, rated for use with hydrogen and optimise vacuum to the best levels possible
- Confirm if a source change or source component change is required
- Ensure a proper conditioning routine is undertaken
- Use method translation software to ensure peak retention order is maintained and check peak identities to confirm
- Be alert to the possibility of ion molecule reactions and investigate / mitigate where necessary
- Confirm spectral integrity and consider impact on quantitative ion ratios in SIM mode
- Verify tuning compatibility