20 Sep 2017
5 ways to improve your Split / Splitless Injection
Split / splitless injectors typically vaporise a sample dissolved in a suitable organic solvent under increased temperature. The sample vapours are entrained into the carrier gas flow inside a ‘liner’ or ‘sleeve’ within the inlet and from there pass into the column or out of the inlet via ‘split’ line / valve.
Splitless injection is used to perform an ‘on-instrument’ dilution and the relative amounts of sample which enter the column or discarded to waste via the split line are adjusted using the relative carrier and split flow rate ratio. See point 3 for why we use split injection and don’t just dilute the sample further prior to injection! Split injection is used when we have enough sample to make up a solution concentrated enough to achieve the required detection limits when only a small portion will reach the column.
In splitless injection the split valve is initially closed, ensuring that all sample passes into the column and is used when sample is limited or analyte concentration is expected to be very low.
Septa are plasticised rubber disks, held under torque, which form a seal around the injection syringe needle so that pressure is maintained during sample introduction. They need to be highly ‘plastic’ in order to do this effectively and therefore many have plasticisers added. These components leach out the septum and can give rise to noisy baselines and, under gradient temperature programming conditions, discrete interference peaks. The septum purge gas flows over the underside of the septum to carry away these bleed products and results in better baselines, fewer ‘unknown’ peaks and better detection limits.
When was the last time you checked the septum purge flow settings or verified the flow via your instrument EPC system or even better with an electronic flow meter attached to the septum purge outlet. You’d be amazed at how many systems have blocked septum purge lines which contribute to lower detection limits or noisy baselines and which go largely undiscovered!
Septa perish over time and become cored - releasing small shards into the inlet which lead to the problems outlined above but which the septum purge flow cannot mitigate. Don’t leave changing your septum until the system won’t hold pressure – change regularly to avoid reactive maintenance and poor chromatography. Further – most septa are held under torque using a nut arrangement – please follow manufacturers guidelines to the level of torque required – too much will mean shorter septum lifetime – too little will result in a leaky system! Septa are rated to different upper temperature limits and some are faced with a thin layer of PTFE to restrict the outgassing of potential contaminants.
So, why do we need split injection, why perform an on-instrument dilution rather than just diluting the sample further, prior to injection?
Firstly, capillary GC columns are limited to the amount of each analyte which can be introduced into the column before peak shapes begin to deteriorate. This is known as sample loading capacity. Smaller i.d. columns and thinner stationary phase films have lower capacity and analyte concentrations in the order of a few nanograms on column are typical.
So – we need to have a reasonably dilute sample. Performing an on-instrument dilution is preferable as it results in sharper (more efficient) peaks. The flow of carrier through the liner is higher in split injection, which carries the sample into the column more quickly (i.e. in a narrower band) and therefore results in less analyte (peak) dispersion in the inlet. The higher the split flow, the faster the analyte is carried onto the column, the sharper are the peaks. Of course, method detection limits should be considered when increasing split flows as sample concentration on column will be reduced and the gain in sensitivity due to the increased peak signal to noise ratio may be outweighed by the reduction in absolute sample amount on-column.
In an experiment where 1ul of solvent is introduced which expands to give 1mL of sample vapour in the inlet, a split flow of 100 mL/min. and a carrier flow of 1mL/min, the split ratio is approximately 100:1. This would mean, in most common instrument designs, a flow through the liner (containing the sample) of 101 mL/min. Our 1 mL of sample vapour would therefore be transported into the column in 1/101 x 60 seconds - approximately 0.6 seconds (a very good initial peak width on column I’m sure you will agree!). When the split ratio is reduced to 10:1, the sample introduction would take 1/11 x 60 seconds – approximately 6 seconds – mmmmmm not so good, but obviously still OK for most applications.
The full explanations of the following rules are outside the scope of this piece – however the rules themselves are vital to getting the best detection limits and quantitative performance from your equipment.
Initial oven temperature setting - should be 10 to 15oC lower than the boiling point of the solvent used to dissolve the sample (sample solvent). In splitless injection the sample vapour moves very slowly into the column and we need to have a mechanism to re-focus the analyte band to avoid very low peak efficiencies. Having a low initial oven temperature traps (focuses) the analyte as it enters the column – sharpening up the analyte band (chromatographic peak) and improving the peak shape and efficiency.
Optimise the splitless time – at some point after injection the split line valve MUST be opened to eject lingering components from the inlet, which would otherwise lead to oversize and tailing solvent peaks and rising baselines throughout the analysis. The splitless time should be determined by monitoring the peak area of analytes at the beginning middle and end of the chromatogram and plotting peak area versus splitless time. Try values in 10 second increments from 10 seconds after injection (10, 20, 30 secondss. etc.) and chose a value which shows a repeatable peak area to the previous time point for the best compromise between chromatographic peak shape and area reproducibility
Match the sample solvent and column polarity – in order to avoid very broad or even split / shouldered peaks. Hexane with 100% PDMS (non-polar) column or Methanol with a Wax (polar) column for example
Avoid backflash – excess sample vapour will contaminate your system and cause carry-over and peak area irreproducibility. Use a backflash calculator to ensure that the volume of sample vapour you create is lower than the available volume within the inlet liner to avoid these issues.
Choice of liner type and packing material is important to ensure good injection reproducibility. Again the underlying theory is too detailed for this piece (see www.chromacademy.com for more details), however there are several aspects that need to be considered which are considered below
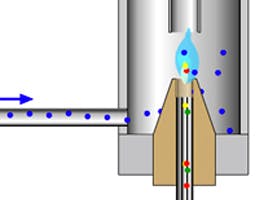
Glass wool packing – deactivated glass wool is used to provide a large surface area from which less volatile components may volatilise, it provides a tortuous route through the liner so that the sample vapour is properly mixed to ensure good homogeneity and also serves to wipe the tip of the syringe to avoid the discrimination processes whereby higher boiling analytes are selectively removed from the inlet prior to them volatilising. For all these reasons, deactivated glass wool in the right amount, density and positioning within the inlet liner can vastly improve the precision and accuracy of sample introduction, especially in splitless injection. If you are unsure of the right amount or positioning of the glass wool please contact your manufacturer.
Liner features – a gooseneck restriction at the bottom of the liner prevents analytes encountering hot metal surfaces and potentially degrading, which is especially useful in splitless analysis. A gooseneck at the top of the liner prevents overfilling of the liner (backflash) and may improve analytical reproducibility.
For split injection, the default choice is a straight through (no gooseneck) unpacked liner. For splitless injection the default choice is a packed liner with a lower gooseneck.