03 Oct 2017
Polar analyte retention and phase collapse
Why would one want to use a mobile phase containing 100% water? Usually to wash the analyte or old mobile phase from the column (stationary phase surface) or to help retain analytes which elute quickly using mobile phases containing only very small amounts of organic solvent (usually highly polar analytes).
It’s fair to say that the use of 100% water eluent or wash solutions should be avoided with hydrophobic (reversed phase) stationary phases to avoid something that we used to call ‘phase collapse’ and which most practitioners now call ‘dewetting’.
The vast majority of an HPLC support particle surface area is contained within the pore structure and that, for a well manufactured phase, the surface in these pores will be covered with hydrophobic ligands (C18, C8 etc.). Whilst the liquid within the pores contains some organic solvent (at least 5% organic is a good rule of thumb), the liquid and hydrophobic surface can live reasonably well together (Figure 1a).
If we switch to a 100% aqueous solvent, whilst under pump pressure, the water will be forced into the pores of the packing material particles, however as the bonded phase on the surface is so highly non-polar, water will not be allowed to penetrate (Figure 1b). As soon as the pressure is released (pump switched off or pressure pulse during injection), then the water will be forced out of the pore due to the repulsive effect of the stationary phase surface (Figure 1c). After this ‘dewetting’ has occurred, even switching the pump back on will not allow water to penetrate the pore and so we will have a situation in which retention will be variable and much shorter than anticipated.
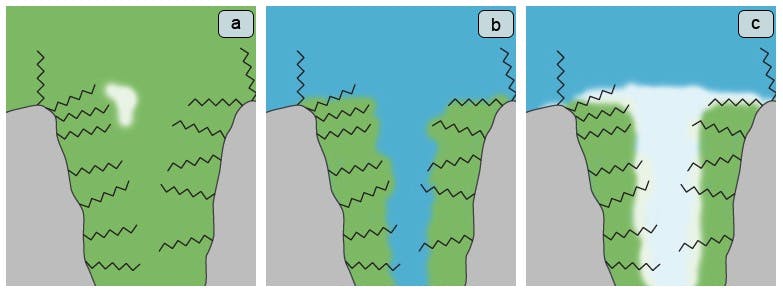
Figure 1
- Inner pore structure and stationary containing eluent with at least 5% organic
- 100% aqueous solvent introduced under pressure - penetrates the pore structure but not the stationary phase
- Pressure released and the highly hydrophobic internal environment of the pore repels the aqeous solution and the pore de-wets
The solutions is to wash the column with an eluent containing at least 5% organic solvent for an extended period of time (usually at increased pressure or flow) to ensure the new eluent system reaches all of the dewetted surface within the pore structures. We recommend a 10% solution of acetonitrile or methanol at the highest pressure you are happy to operate at within the safe operating constraints of the both pump and column, for at least 20 column volumes.
This doesn’t get us round the issue of retaining polar analytes however…..
In fact, polar analyte retention and separation is usually achieved using specially adapted surfaces / ligands which have modified to be less hydrophobic or which contain a polar group embedded within them to allow the stationary phase surface to be penetrated by totally aqueous solutions.
Manufacturers will achieve this is a whole variety of different ways – here are just a few from the Element stable of columns:
Incorporate a polar group into the alkyl chain used as the ligand (usally called Polar Embedded columns) examples include Zorbax Bonus RP, a fully water wettable column with a highly unusual (orthogonal) selectivity
Use a slightly polar end-capping reagent which facilitates the adsorption of a layer of water at the silica surface, examples include YMC Hydrosphere which is specifically designed for 100% aqueous applications
Control the amount of ligand applied to the silica surface in combination with a very inert silica surface (usually called a Type B silica) which allows the silanol groups to adsorb a layer of water in a similar fashion to that described above, examples here include the excellent ‘Optimal L’ columns which give a highly robust and lower cost option for polar analyte retention
HILIC columns can also be used for polar analyte retention through the use of polar stationary phase ligands used in conjunction with highly organic eluent systems
Mixed mode columns are most effective as they use ligands which incoportae both hydrophic and ionisable groups which can be ‘tuned’ for analyte retention through the use ofg eluent pH and buffers and produce superb separations for polar and iponisable analytes
A very new approach to the retention and separation of analytes with polar functional groups is Specific Hydrogen-bond Adsorption Resolution Column (SHARC) chromatography, which uses specific hydrogen bonding interactions in tandem with fully organic mobile phases to produce highly selective separations very quickly.